CAR-T vs CAR-NK Cell: The Cancer Cell Therapy Showdown (2026)
One of the most promising approaches in cellular therapies is immunotherapies using engineered T cells or natural killer (NK) cells expressing chimeric antigen receptors (CARs). Will CAR-NK cell therapies overshadow traditional CAR-T options for the treatment of some cancers?
Recently researchers have tried to modify natural killer (NK) cells with chimeric antigen receptors (CAR) to increase potential to bind and destroy cancer cells, known as NK cell therapy. Here we describe the differences between CAR T cell therapies and CAR NK cell therapies to consider when developing cancer treatments for patients.
There are multiple CAR T-cell products in various stages of approval and development that are available. Ciltacabtagene autoleucel (cilta-cel; Carvykti, Janssen Biotech, Inc/Legend Biotech) is particularly notable for its efficacy, as demonstrated in the CARTITUDE-1 trial (NCT03548207). Patients with MM received a single infusion of cilta-cel and achieved remission without the need for maintenance therapy. These outcomes imply the curative potential of CAR T-cell therapy, further establishing its critical role in the treatment landscape for MM and other hematologic malignancies. (3)
As of April 2026, there are 7 FDA approved CAR T-cell therapies and multiple other investigational products (Table). However, despite their significant efficacy, challenges for implementation and operationalizing these therapies remains a persistent challenge across clinical settings. (3)
While CAR T-cell therapy is more well-known and studied, there are challenges to the manufacturing of CAR T that does not exist with CAR-NK. This may prove to be a game-changer for blood cancer patients who do not have the time to wait for a CAR T-cell donor or for their own cells to be engineered for treatment.

At least in my mind, potentially the advantages could be the safety profile of CAR-NK cells. Of course, we have treated only a small number of patients and our follow-up is certainly shorter than follow-ups with patients who have been treated with CAR T-cells. But in this small group of patients, at least, we haven't seen the toxicities that had been described with the CAR T-cells, such as the cytokine storm or the neurological complications. And none of our patients for instance required any support on the intensive care unit, which many patients with CAR T-cells do.
Now, the other unique aspect of natural killer cells is unlike T-cells, natural killer cells when taken from source that is unrelated to the patient, will not cause graft versus host disease. So, natural killer cells do not have the capability to pose graft versus host disease. It's to do with their biology. And this opens the way then to go and make banks of natural killer cells from a donor that could be completely unrelated to a patient and to make these biobanks themselves, that then can be used to treat patients pretty much off the shelf.


If we are capable, if we're able to have an off the shelf product so that the patient comes into your office and without any consideration for matching, without any consideration for growing, you have these cells frozen, they're sitting in a biobank and you can thaw and infuse that would make the strategy obviously much more attractive and also cheaper because if you can make multiple products from one donor, you bring the cost of manufacturing and the cost of therapy.
Will CAR-NK Cell Therapy Become Widely Available?
You need to have a source of cell. You need to have a way of reliably freezing the cells so that you're certain that when you thaw the cells, they are as functional and as effective as the fresh counterpart. And that's what we are working on at the moment. In the study that we've been doing so far, the CAR-NK cells are actually manufactured fresh and given to the patients fresh. We've so far been using one umbilical cord unit to manufacture CAR-NK cells for one patient and to infuse into that patient. But our next steps are now to manufacture multiple doses of CAR-NK cells that then we can freeze and to infuse into patients.
Recently researchers have tried to modify natural killer (NK) cells with chimeric antigen receptors (CAR) to increase potential to bind and destroy cancer cells, known as NK cell therapy. Here we describe the differences between CAR T cell therapies and CAR NK cell therapies to consider when developing cancer treatments for patients.
What is CAR-T?
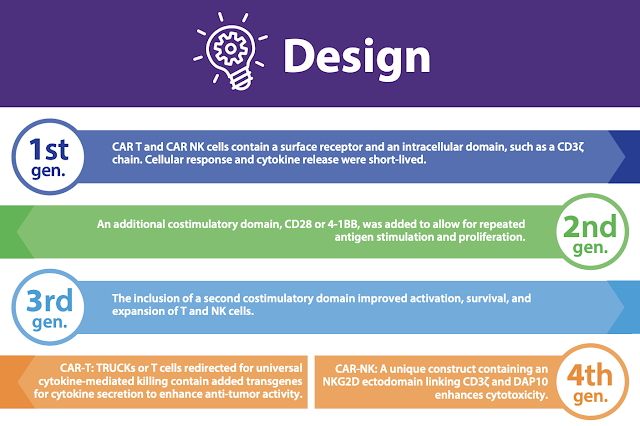
T cells are part of the adaptive immune system and are intended to generate a long lasting protective immune response. By engineering T cells with the Natural Killer Group 2D (NKG2D) receptor, they become NKG2D CAR T cell therapies.
Chimeric antigen receptor (CAR) T-cell therapy demonstrates significant, clinically meaningful outcomes for patients with hematologic malignancies, such as B-cell lymphomas or multiple myeloma (MM). These novel therapies are at the helm of innovation and the evolution of cellular immunotherapies. At the 2026 Community Oncology Alliance meeting, experts discussed the current landscape for CAR T-cell treatment and how clinics, cancer centers, and other points of care can expand access to these efficacious therapies to patients who need them.
CAR T-cell therapy is an innovative approach to utilizing a patient’s own immune system to attack cancer cells. The process involves harvesting the immune cells and manufacturing them to target specific antigens or biomarkers on the surface of malignant cells. Emerging data from research and clinical trials show improved response rates and, notably, minimal residual disease (MRD) negativity. Over the past few years, and per the updated International Myeloma Working Group, MRD plays a central role in assessing treatment response. (1,2)
There are multiple CAR T-cell products in various stages of approval and development that are available. Ciltacabtagene autoleucel (cilta-cel; Carvykti, Janssen Biotech, Inc/Legend Biotech) is particularly notable for its efficacy, as demonstrated in the CARTITUDE-1 trial (NCT03548207). Patients with MM received a single infusion of cilta-cel and achieved remission without the need for maintenance therapy. These outcomes imply the curative potential of CAR T-cell therapy, further establishing its critical role in the treatment landscape for MM and other hematologic malignancies. (3)
As of April 2026, there are 7 FDA approved CAR T-cell therapies and multiple other investigational products (Table). However, despite their significant efficacy, challenges for implementation and operationalizing these therapies remains a persistent challenge across clinical settings. (3)
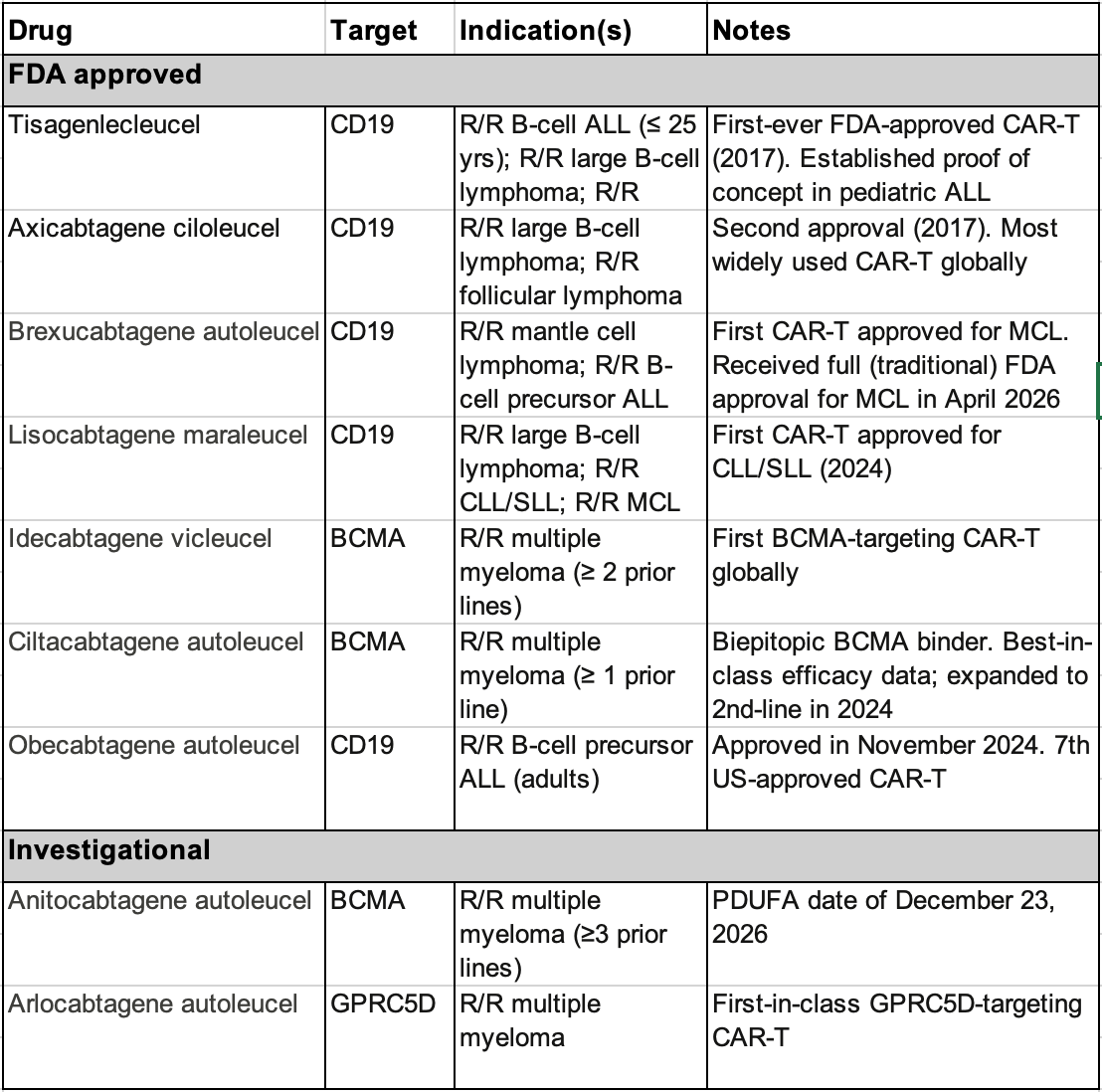 |
Note: ALL, acute lymphocytic leukemia; MCL, mantle cell lymphoma; CLL/SLL, chronic lymphocytic leukemia/small lymphocytic leukemia What is CAR-NK? |
NK cells are a part of the innate immune system and are the first line of defense against foreign infections and cancer cells. Their primary role is surveillance, early detection of foreign cells and initial immune system activity. By engineering NK cells with a CAR receptor, they become CAR NK cell therapies.
CAR-T vs CAR-NK

According to Dr. Rezvani, who is a researcher at MD Anderson in Houston:
At least in my mind, potentially the advantages could be the safety profile of CAR-NK cells. Of course, we have treated only a small number of patients and our follow-up is certainly shorter than follow-ups with patients who have been treated with CAR T-cells. But in this small group of patients, at least, we haven't seen the toxicities that had been described with the CAR T-cells, such as the cytokine storm or the neurological complications. And none of our patients for instance required any support on the intensive care unit, which many patients with CAR T-cells do.
Now, the other unique aspect of natural killer cells is unlike T-cells, natural killer cells when taken from source that is unrelated to the patient, will not cause graft versus host disease. So, natural killer cells do not have the capability to pose graft versus host disease. It's to do with their biology. And this opens the way then to go and make banks of natural killer cells from a donor that could be completely unrelated to a patient and to make these biobanks themselves, that then can be used to treat patients pretty much off the shelf.

How are CAR T-Cells Made?
Currently, the way CAR T-cells are made is, at least for the commercially available CAR T-cells, you have to collect the blood from the patient. You have to manufacture the CAR T-cells and then give it back to the patient. And that brings two major points that we need to bear in mind. One is the cost and the logistics and also with some patients who have very aggressive disease, they may not have the four weeks or six weeks that is needed.
If we are capable, if we're able to have an off the shelf product so that the patient comes into your office and without any consideration for matching, without any consideration for growing, you have these cells frozen, they're sitting in a biobank and you can thaw and infuse that would make the strategy obviously much more attractive and also cheaper because if you can make multiple products from one donor, you bring the cost of manufacturing and the cost of therapy.
Will CAR-NK Cell Therapy Become Widely Available?
You need to have a source of cell. You need to have a way of reliably freezing the cells so that you're certain that when you thaw the cells, they are as functional and as effective as the fresh counterpart. And that's what we are working on at the moment. In the study that we've been doing so far, the CAR-NK cells are actually manufactured fresh and given to the patients fresh. We've so far been using one umbilical cord unit to manufacture CAR-NK cells for one patient and to infuse into that patient. But our next steps are now to manufacture multiple doses of CAR-NK cells that then we can freeze and to infuse into patients.
How do Chimeric Antigen Receptor (CAR) Therapies and Stem Cell Transplants Compare?
According to Dr. Rezvani:
I think what is happening is that the field of stem cell transplantation is evolving. And for certain types of cancers, it is very possible that CAR T-cells will, or CAR-NK cells will supersede allogeneic stem cell transplantation. For other types of cancers, probably stem cell transplantation will stay with us for a while longer. Ultimately, I think the goal is to be able to provide our patients with the most effective therapy with the least amount of toxicity. And that's where I'm hoping and I pray that we will be heading towards. At least for lymphomas and the lymphoid malignancies, I really do believe that the field of stem cell transplantation is already in the evolution of going towards cell therapy.
I think what is happening is that the field of stem cell transplantation is evolving. And for certain types of cancers, it is very possible that CAR T-cells will, or CAR-NK cells will supersede allogeneic stem cell transplantation. For other types of cancers, probably stem cell transplantation will stay with us for a while longer. Ultimately, I think the goal is to be able to provide our patients with the most effective therapy with the least amount of toxicity. And that's where I'm hoping and I pray that we will be heading towards. At least for lymphomas and the lymphoid malignancies, I really do believe that the field of stem cell transplantation is already in the evolution of going towards cell therapy.
Patient Dies From Rare Cancer Linked to CAR T-Cell Therapy–What Are the Risks?
A patient developed a rare and fatal form of blood cancer just one month after receiving chimeric antigen receptor (CAR) T-cell therapy, according to a 2024 case study published in the New England Journal of Medicine.
The authors wrote that CAR T-cell therapy, which treats cancer by modifying the genes of a patient’s immune cells, did not cause the T-cell lymphoma.
CAR T-cell therapy has improved the prognosis for several blood-related cancers by utilizing patients’ own immune cells to fight cancer more effectively. However, as the treatment becomes more widespread, concerns are growing over rare but serious adverse effects, including the development of secondary cancers.
The authors wrote that CAR T-cell therapy, which treats cancer by modifying the genes of a patient’s immune cells, did not cause the T-cell lymphoma.
CAR T-cell therapy has improved the prognosis for several blood-related cancers by utilizing patients’ own immune cells to fight cancer more effectively. However, as the treatment becomes more widespread, concerns are growing over rare but serious adverse effects, including the development of secondary cancers.
References:
Landgren O, Kazandjian D. MRD and Plasma Cell Dynamics after CAR T-cell Therapy in Myeloma. Blood Cancer Discov. 2023;4(5):346-348. doi:10.1158/2643-3230.BCD-23-0134
Kumar S, Paiva B, Anderson KC, et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 2016;17(8):e328-e346. doi:10.1016/S1470-2045(16)30206-6
- Boccia R, Byrne M, Patel A, et al. CAR T Comes to Main Street: Expanding Access, Empowering Care. Presented at: 2026 COA Annual Meeting. April 28-29, 2026. Orlando, FL.
- https://www.patientpower.info/acute-lymphoblastic-leukemia/emerging-research/comparing-car-nk-and-car-t-cell-therapies
- https://www.biolegend.com/en-us/car-t-infographic
- https://celyad.com/wp-content/uploads/2021/04/CAR-T-vs.-NK-cells_Fact-Sheet.pdf
- https://www.theepochtimes.com/health/patient-dies-from-rare-cancer-linked-to-car-t-cell-therapy-what-are-the-risks-5733839
- CAR-T vs CAR-NK Therapy — and Why Metabolic Modulation May Decide the Winner (2026)


.png)





.png)
.png)
.png)
Comments
Post a Comment